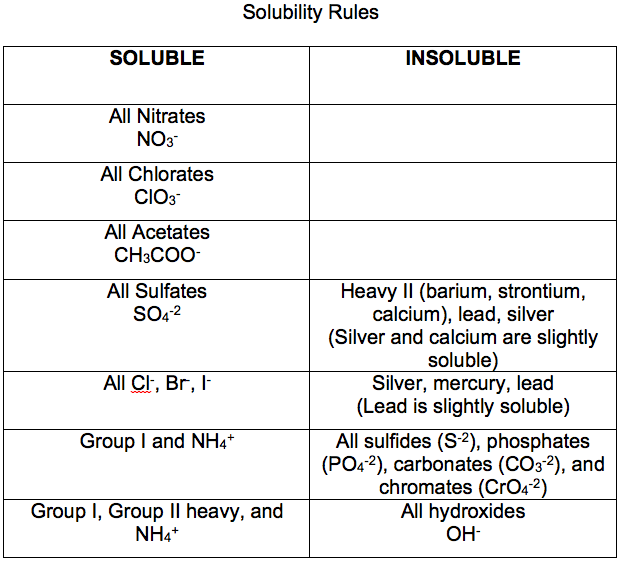
 Therefore, this compound will not contribute to any precipitation being formed. Although Rule #5 says that hydroxides tend to be insoluble, Rule #1 states that salts of alkali metal cations tend to be soluble, and Rule #1 precedes Rule #5. The first product, KOH, is an example of two rules contradicting each other. Predict if a precipitate will form as a result of this reaction:Ĭonsider again the products of the reaction: if either is insoluble, a precipitate will form. Therefore, because of this compound, a precipitate will form in the course of this reaction.ĥ. According to Rule #5, that sulfides tend to be insoluble. A precipitate of this compound will not form. The products of the reaction must be examined if either of the substances formed in the reaction is insoluble, a precipitate will form.Ĭonsidering NaNO 3, Rule #3 states that nitrates tend to be soluble. Predict whether a precipitate will form as a result of this reaction: Because Rule #3 precedes Rule #4, the compound is insoluble and will form a precipitate.Ĥ. Rule #4 states that bromides are usually soluble, but Rule #3 states that salts of silver are insoluble. Letters a and c are both likely to form precipitates.Ĭoncerning a) CaSO 4, although sulfates tend to be soluble, Rule #5 indicates that calcium sulfate is an important exception to this rule.įor b), Rule #1 indicates that table salt (NaCl) is soluble because it is a salt of an alkali metal.Ĭ) is an example of two rules contradicting each other. Which of these substances is likely to form a precipitate? Therefore, it will not form a precipitate.ģ. This is perchlorate, which according to Rule #2 is likely to be soluble. Therefore, FeCO 3 is likely to form a precipitate.Ģ. Fluorides such as BaF 2, MgF 2, and PbF 2 are frequently insoluble.Īccording to Rule #5, carbonates tend to be insoluble. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
